Sickle Cell Disease
Sickle cell disease (SCD) is a genetic blood disorder that affects the protein hemoglobin found inside red blood cells. Hemoglobin carries oxygen from the lungs to the rest of the body (OpenStax 2019). One of the most common symptoms is painful episodes called sickle cell crises, which occur when misshapen cells block blood flow in small vessels. Patients often experience chronic anemia, resulting in fatigue, weakness, and shortness of breath. Other symptoms include swelling in the hands and feet, frequent infections, delayed growth in children, and vision problems. Over time, repeated blockage of blood vessels can cause organ damage, particularly in the spleen, lungs, and brain (National Heart, Lung, and Blood Institute 2022; Rees et al. 2010).
In people with sickle cell disease, a mutation in the beta-globin gene results in the production of an abnormal type of hemoglobin called hemoglobin S. The hemoglobin S proteins stick together and form long chains, which distort red blood cells into a crescent or “sickle” shape, especially at low oxygen concentrations (OpenStax 2019).
Unlike normal red blood cells, which are flexible and round, sickled cells are:
- Rigid
- Sticky
- More fragile
Because of their shape, sickled cells can block small blood vessels because they are unable to squeeze through. This can block blood flow and reduce oxygen delivery to tissues causing severe pain episodes called sickle cell crises.
Other complications may include:
- Chronic anemia
- Organ damage
- Increased risk of infection
- Reduced life expectancy without treatment

Sickle Cell Trait
Not everyone who inherits the sickle cell gene develops the disease.
Individuals who inherit one sickle cell allele and one normal allele have sickle cell trait. These individuals usually do not experience symptoms because most of their hemoglobin is normal.
However, they can pass the sickle cell allele to their children.
Malaria and Sickle Cell: An Evolutionary Connection
One of the most well-known examples of natural selection in humans involves the relationship between sickle cell trait and malaria (Hedrick 2011).
Malaria is a parasitic infection that targets red blood cells and often begins with flu-like symptoms. The most characteristic feature is cyclical fever, where patients experience repeated episodes of fever, chills, and sweating as the parasite reproduces in the blood. Other common symptoms include fatigue, headache, muscle aches, and nausea, which result from the body’s immune response and red blood cell destruction. As the infection progresses, patients may develop anemia and jaundice due to the breakdown of red blood cells. In severe cases, malaria can lead to confusion, difficulty breathing, organ failure, and death if not treated promptly (World Health Organization 2021; Kotepui et al. 2015).
In regions where malaria is common, particularly sub-Saharan Africa, people with sickle cell trait have a survival advantage. Red blood cells containing sickle hemoglobin make it more difficult for the Plasmodium malaria parasite to reproduce (Gong et al. 2013). Altered hemoglobin, increased cellular stress, and early removal of infected cells by the spleen interfere with the parasite’s life cycle.
Because individuals with sickle cell trait are more resistant to severe malaria, the sickle cell allele has remained common in populations from malaria-endemic regions.
This phenomenon is known as balanced polymorphism, in which a genetic trait persists in a population because it provides a survival advantage under certain environmental conditions.
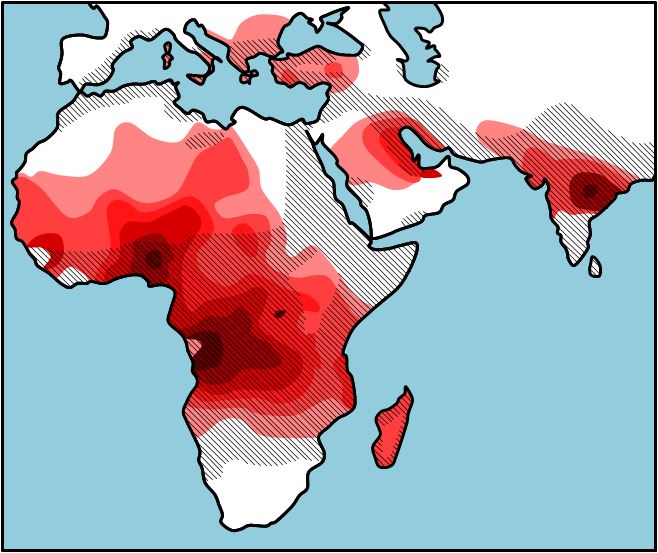
Figure 4.“The hatched line represents the distribution of malaria. The various red colors represent the relative frequency of sickle cell allele in the population with the dark red having the highest frequency and the light red having the lowest frequency. Work by Eva Horne.“https://bio.libretexts.org/Courses/University_of_California_Davis/BIS_2A%3A_Introductory_Biology_-_Molecules_to_Cell/BIS_2A%3A_Introductory_Biology_2018_(Singer)/MASTER_RESOURCES/Sickle_Cell_Anemia*
Did You Know?
People with sickle cell trait often have increased protection against severe malaria, which is why the sickle cell gene is more common in regions where malaria has historically been widespread.
This is one of the most widely cited examples of natural selection in human populations.
Equity, Diversity, and Inclusion (EDI) in Sickle Cell Disease
Sickle cell disease is an important example of how genetics, ancestry, healthcare access, and systemic inequities intersect in medicine.
- Ancestry and Misconceptions:
Sickle cell disease is most commonly associated with individuals whose ancestors come from:
- Sub-Saharan Africa
- The Mediterranean
- The Middle East
- India
- Parts of South and Central America
However, it is important to understand that sickle cell disease is not tied to race, but rather to geographic regions where malaria historically occurred.
This distinction highlights the difference between biological ancestry and socially constructed ideas of race. (Centers for Disease Control and Prevention 2023; National Heart, Lung, and Blood Institute 2022)
- Historical Bias in Research and Funding
Despite being one of the most common inherited blood disorders in the world, sickle cell disease has historically received less research funding and medical attention compared to other genetic diseases.
For example, diseases that primarily affect populations of European ancestry, such as cystic fibrosis, have historically received significantly more funding per patient in biomedical research (Farooq et al. 2020; National Institutes of Health 2022).
These disparities reflect broader patterns of systemic inequities in healthcare research and funding.
- Barriers in Healthcare
Many individuals living with sickle cell disease face challenges within healthcare systems, including:
- Delayed diagnosis
- Limited access to specialized treatment
- Under-treatment of pain
- Healthcare bias and stigma
Patients experiencing sickle cell crises often require urgent pain management, but research has shown that some patients report not being taken seriously when seeking care.
Addressing these issues requires improving:
- Medical education
- Cultural competency in healthcare
- Access to specialized care
(Lanzkron et al. 2013; Tanabe et al. 2019)
- Representation and Awareness
Increasing awareness of sickle cell disease is an important step toward improving patient outcomes. Despite scientific advances, researchers emphasize that challenges remain. Gene‑editing therapies work to reduce sickling (Frangoul et al. 2021); however, they are complex, expensive, and currently available only at specialized treatment centers. Because these therapies are new, patients must be followed for many years to ensure the treatment remains effective and does not cause unexpected long‑term side effects. Ethical considerations and equitable access are also central to ongoing research discussions, particularly given the global burden of sickle cell disease in regions with limited healthcare infrastructure (Subica 2023; Patel and Kuo 2025).
Public health initiatives now focus on:
- Newborn screening programs
- Genetic counseling
- Community education
- Research into new treatments, including gene therapy
These efforts help promote health equity and ensure that patients with sickle cell disease receive appropriate care and support. (Centers for Disease Control and Prevention 2023; World Health Organization 2021; National Heart, Lung, and Blood Institute 2022).
Practice Activity
References
CDC. 2024 Apr 22. Sickle Cell Disease (SCD). Sickle Cell Disease (SCD). https://www.cdc.gov/sickle-cell/index.html.
Centers for Disease Control and Prevention. 2023. Sickle cell disease (SCD) [Internet]. Atlanta (GA): CDC; [cited 2026 May 3]. Available from: https://www.cdc.gov/sicklecell/
Farooq F, Mogayzel PJ, Lanzkron S, Haywood C Jr, Strouse JJ. 2020. Comparison of US federal funding for sickle cell disease and cystic fibrosis. JAMA Netw Open. 3(3):e201737.
Frangoul H, Altshuler D, Cappellini MD, et al. 2021. CRISPR–Cas9 gene editing for sickle cell disease. N Engl J Med. 384(3):252–260.
Gong L, Parikh S, Rosenthal PJ, Greenhouse B. 2013. Biochemical and immunological mechanisms by which sickle cell trait protects against malaria. Malar J. 12:317.
Hedrick PW. 2011. Population genetics of malaria resistance in humans. Heredity. 107(4):283–304.
Kotepui M, Piwkham D, PhunPhuech B, Phiwklam N, Chupeerach C, Duangmano S. 2015. Effects of malaria parasite density on blood cell parameters. Infect Dis Poverty. 4:45.
Lanzkron S, Carroll CP, Haywood C Jr. 2013. Mortality rates and age at death from sickle cell disease: US, 1979–2005. Public Health Rep. 128(2):110–116.
National Heart, Lung, and Blood Institute. 2022. Sickle cell disease [Internet]. Bethesda (MD): NIH; [cited 2026 May 3]. Available from: https://www.nhlbi.nih.gov/health/sickle-cell-disease
National Institutes of Health. 2022. Estimates of funding for various research, condition, and disease categories (RCDC) [Internet]. Bethesda (MD): NIH; [cited 2026 May 3]. Available from: https://report.nih.gov/funding/categorical-spending#/
National Heart, Lung, and Blood Institute. 2022. Sickle cell disease [Internet]. Bethesda (MD): NIH; [cited 2026 May 3]. Available from: https://www.nhlbi.nih.gov/health/sickle-cell-disease
OpenStax. 2019. Anatomy and physiology 2e. Houston (TX): OpenStax, Rice University. Section 18.3, Erythrocytes: sickle cell anemia. https://openstax.org/details/books/anatomy-and-physiology-2e
Patel A, Kuo A. 2025. The ethics of gene therapy for sickle cell disease. Cureus. 17(3):e81037.
Rees DC, Williams TN, Gladwin MT. 2010. Sickle-cell disease. Lancet. 376(9757):2018–2031.
Subica AM. 2023. CRISPR in public health: the health equity implications and role of community in gene-editing research and applications. Am J Public Health. 113(8):874–882.
Tanabe P, Spratling R, Smith D, Grissom P, Hulihan M. 2019. CE: understanding the complications of sickle cell disease. Am J Nurs. 119(6):26–35.
World Health Organization. 2021. Sickle-cell disease [Internet]. Geneva (Switzerland): WHO; [cited 2026 May 3]. Available from: https://www.who.int/news-room/fact-sheets/detail/sickle-cell-disease
World Health Organization. 2021. Malaria [Internet]. Geneva (Switzerland): WHO; [cited 2026 May 3]. Available from: https://www.who.int/news-room/fact-sheets/detail/malaria